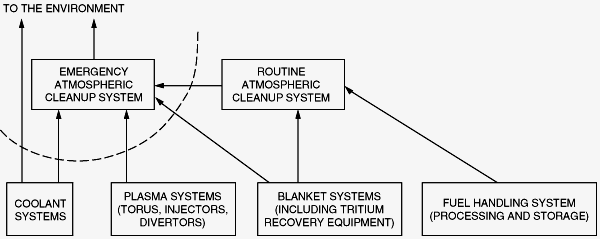


Cleanup systems are required to protect the environment from accidental
and routine releases of tritium.

What are the underlying factors that change tritium gas, T2, to tritiated water, HTO?

Tritium will be a fuel at future fusion reactors, but health risks for tritium differ between its form as T2 gas and its form as HTO. Because tritium in the water form is about 25,000 times more hazardous than in the gas form, evaluation of human health risks for a number of situations around future fusion reactor plants depends on knowledge of how tritium converts from T2 to HTO. A series of experimental and theoretical studies were undertaken to measure needed parameters and to calculate others. This effort resulted in the development of validated rate equations, which spanned more than seven orders of magnitude in tritium concentration. Health risks from a wide variety of possible situations can now be calculated. These calculations provide input for future design and siting requirements for tritium-fueled fusion reactors.

Jacobs, D. G., C. E. Easterly, and J. E. Phillips. Influence of the rate of conversion of HT to HTO on projected radiation doses from release of molecular tritium, pp. 373-383. In Proceedings of the IAEA Symposium on the Behavior of Tritium in the Environment, October 16-20, 1978, San Francisco, Calif.
Easterly, C. E. and M. R. Bennett. 1983. Radiation catalyzed conversion of tritium gas to tritiated water, Nuclear Technology/Fusion 4(2):116-120.
Noguchi, H., C. E. Easterly, and M. R. Bennett. 1989. Conversion of low-concentration tritium water, Fusion Technology 16(2):137-142.
Easterly, C. E., G. S. Hill, and J. B. Cannon. 1989. Environmental effects of fusion power plants, Part III: Potential radiological impacts of environmental releases, Fusion Technology 16(2):125-136.
Integrated Assessment Briefs. 1995. ORNL/M-4227. Oak Ridge National Laboratory, Oak Ridge, TN.