Biogeochemical Transformations at Critical Interfaces
ORNL Mercury Science Focus Area (SFA)
Featured Research Highlight
Overlooked Mercury Isotope Exchange in Environmental Tracer Studies and Implications
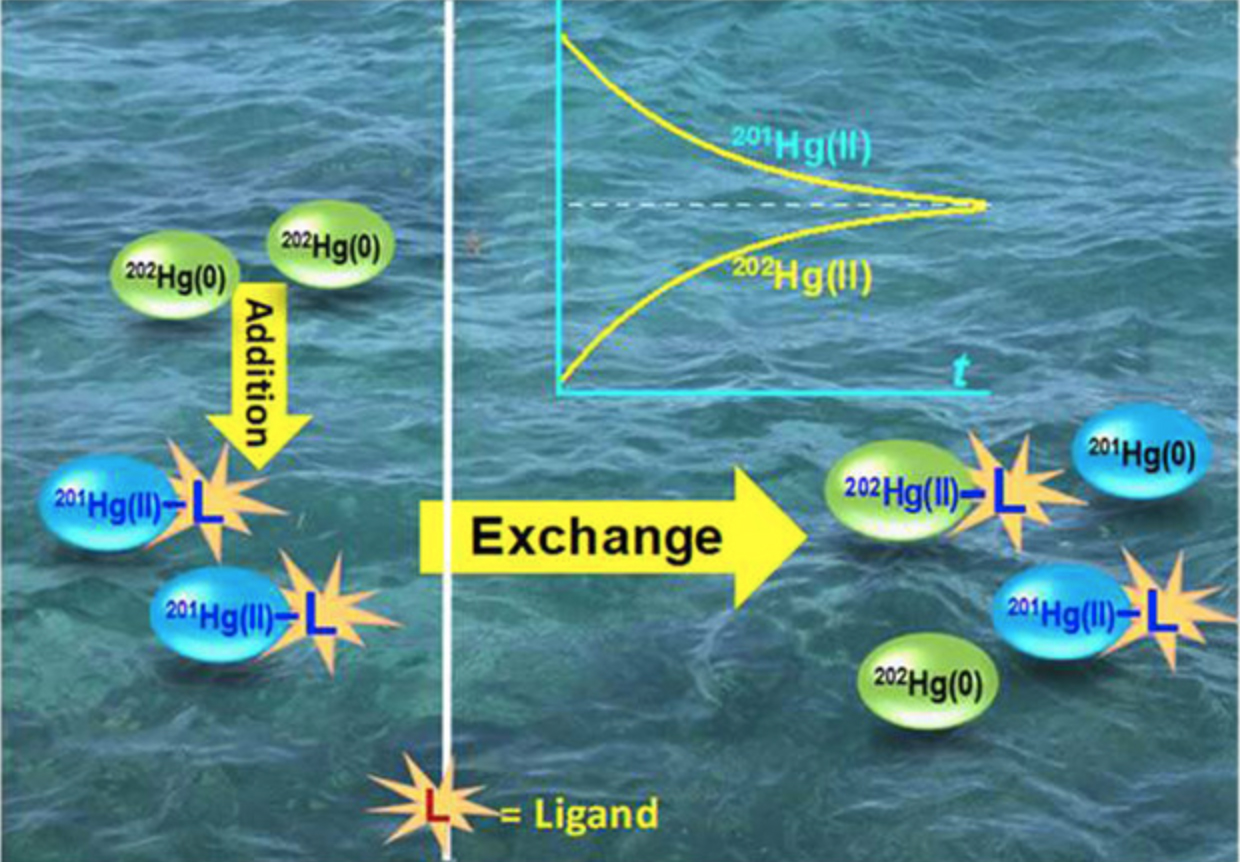
Rapid, spontaneous mercury (Hg) isotope exchange in environmental matrices alter Hg isotope redistribution and thus affects Hg bioavailability and methylmercury (MeHg) production.
The Science
Enriched Hg-stable isotopes have been widely used as tracers in field and laboratory investigations of Hg biogeochemical transformations such as methylation and demethylation. Few studies, however, have considered concurrent isotope exchange reactions between newly spiked and preexisting ambient Hg in environmental matrices, which may alter redistribution and thus transformation of the spiked and ambient Hg. Using enriched Hg [as mercuric Hg(II) or dissolved elemental Hg(0)aq], this study investigated isotope exchange between spiked enriched Hg and ambient Hg(II) bound to various environmental matrices, including soil minerals, low-molecularweight (LMW) thiols, and dissolved organic matter (DOM). The impact of isotope exchange on MeHg production in the presence of organic ligands was also evaluated with an iron-reducing bacterium Geobacter sulfurreducens PCA. Without external reductants or oxidants, the exchange between Hg(0)aq and Hg(II)resulted in transfers of two electrons and redistribution of Hg isotopes bound to the ligand but no net changes of chemical species in the system. Similarly, the spiked Hg(II) rapidly exchanges with ligand- or mineral-bound ambient Hg(II), resulting in redistribution of Hg isotopes bound to the ligands or minerals and an apparently similar methylation rate and magnitude of the spiked Hg and ambient Hg by PCA cells. These observations underscore the importance of isotope exchange when an enriched Hg isotope is applied in environmental matrices, as the exchange could potentially lead to biased rate calculations of Hg transformation and bioaccumulation and thus result in biased risk assessments of new Hg input to the natural ecosystems.
The Impact
The research highlights the importance of considering isotope exchange reactions in experimental enriched Hg tracer studies or in studies where natural abundance Hg isotope are present in environmental systems.
Summary
We investigated the rates and dynamics of Hg isotope exchange between spike-enriched Hg and ambient Hg bound to various environmental matrices, and found surprisingly rapid, spontaneous Hg isotope exchange reactions resulting in redistribution of Hg isotopes and changes in Hg bioavailability. Without consideration of these reactions in enriched Hg tracer studies, the estimated rates and predictions of Hg transformation and bioaccumulation could be biased in environmental systems.
Publications
Wang, Q., L. Zhang, X. Liang, X. Yin, Y. Zhang, W. Zheng, E. M. Pierce, and B. Gu. 2020. “Rates and dynamics of mercury isotope exchange between dissolved elemental Hg(0) and Hg(II) bound to organic and inorganic ligands.” Environmental Science & Technology. 54(23): 15534−45. DOI:10.1021/acs.est.0c06229.
Zhang, L., X. Liang, Q. Wang, Y. Zhang, X. Yin, X. Lu, E. M. Pierce, and B. Gu. 2021. “Isotope exchange between mercuric [Hg(II)] chloride and Hg(II) bound to minerals and thiolate ligands: Implications for enriched isotope tracer studies.” Geochimica et Cosmochimica Acta. 292:468−81. DOI:10.1016/j.gca.2020.10.013.
 ORNL Mercury SFA sponsored by Subsurface Biogeochemical Research (SBR) program, U.S. Department of Energy's Office of Biological and Environmental Research. Paul Bayer, SBR Program Manager.
ORNL Mercury SFA sponsored by Subsurface Biogeochemical Research (SBR) program, U.S. Department of Energy's Office of Biological and Environmental Research. Paul Bayer, SBR Program Manager.
Security Notice | Contact Eric Pierce, ORNL | Website Questions | Site Map
File last modified: Wednesday, September 15, 2021
The ORNL Mercury SFA is sponsored by the Subsurface Biogeochemical Research (SBR) program within the U.S. Department of Energy's Office of Biological and Environmental Research.