Biogeochemical Transformations at Critical Interfaces
ORNL Mercury Science Focus Area (SFA)
Featured Research Highlight
Is Particulate-Bound Mercury Available for Microbial Uptake and Methylation?
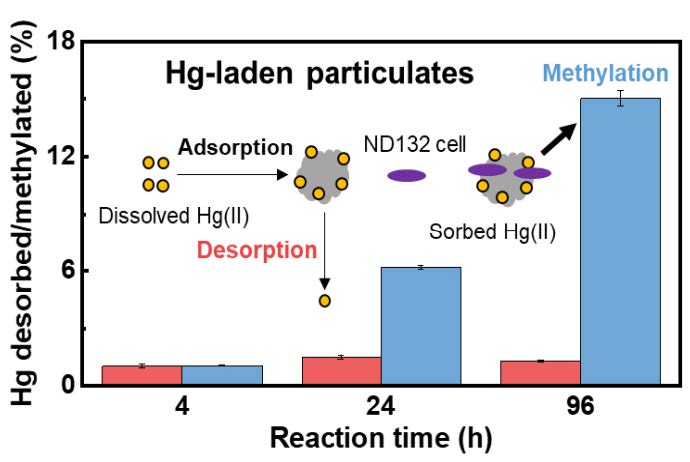
The Hg methylation rate is 4 to 10 times higher than the desorption rate of Hg, suggesting direct contacts and interactions between bacterial cells and the particulate-bound Hg for rapid exchange or uptake.
Study reveals important roles of mineral-adsorbed or particulate-bound mercury (Hg) as a significant source of Hg for microbial methylation in the environment
The Science
In natural freshwater and sediments, mercury (Hg) is largely associated with particulate minerals and organics, but it remains unclear under what conditions particulates may become a sink or a source for Hg and whether particulate-bound Hg is bioavailable for microbial uptake and methylation. We investigate Hg sorption-desorption characteristics on minerals and a Hg-contaminated sediment and evaluate the potential of particulate-bound Hg for microbial methylation. Mercury rapidly adsorbs onto particulates, especially the cysteine-coated hematite and sediment, with little desorption observed (0.1–4%). However, the presence of Hg-binding ligands, such as low-molecular-weight thiols and humic acids, results in up to 60% of Hg(II) desorption from the Hg-laden hematite particulates, but <6% from the sediment. Importantly, the particulate-bound Hg(II) is bioavailable for uptake and methylation by a sulfate-reducing bacterium Desulfovibrio desulfuricans ND132 under anaerobic incubations, and the methylation rate is 4 to 10 times higher than the desorption rate of Hg(II). These observations suggest direct contacts and interactions between bacterial cells and the particulate-bound Hg(II), resulting in rapid exchange or uptake of Hg(II) by the bacteria. Our results highlight the importance of Hg(II) partitioning at particulate-water interfaces and the role of particulates as a significant source of Hg(II) for methylation in the environment.
The Impact
Our study indicates an alternative pathway in which microbes take up Hg that is more complicated than previously thought: Particulate-bound Hg does not have to be desorbed or dissolved to make it available for microbial uptake and methylation.
Summary
Mineral- or particulate-bound Hg is often considered unavailable for microbial uptake and methylation. Our study reveals that particulate-bound Hg(II) is readily available for uptake and methylation by a sulfate-reducing bacterium, D. desulfuricans ND132, under anaerobic incubations and should be considered in predicting methylmercury production in the natural aquatic environment.
Publication
Zhang, L., S. Wu, L. Zhao, X. Lu, E. M. Pierce, and B. Gu. 2019. “Mercury sorption and desorption on organomineral particulates as a source for microbial methylation.” Environmental Science & Technology. 53(5):2426–433. DOI: 10.1021/acs.est.8b06020.
 ORNL Mercury SFA sponsored by Subsurface Biogeochemical Research (SBR) program, U.S. Department of Energy's Office of Biological and Environmental Research. Paul Bayer, SBR Program Manager.
ORNL Mercury SFA sponsored by Subsurface Biogeochemical Research (SBR) program, U.S. Department of Energy's Office of Biological and Environmental Research. Paul Bayer, SBR Program Manager.
Security Notice | Contact Eric Pierce, ORNL | Website Questions | Site Map
File last modified: Thursday, August 08, 2019
The ORNL Mercury SFA is sponsored by the Subsurface Biogeochemical Research (SBR) program within the U.S. Department of Energy's Office of Biological and Environmental Research.